2 min read
Meet Veristat at the ASCO Annual Meeting
May 29 - June 2, 2026 | Chicago, IL
▶️ At the Forefront of Oncology Progress
Veristat is pleased to attend the ASCO...
1 min read
Based on an online seminar presentation made in collaboration with TOPRA and Veristat, this article dives into the future of the European medicines and medical devices industry and the changes in legislation that are being prompted by the evolving pharmaceutical environment. 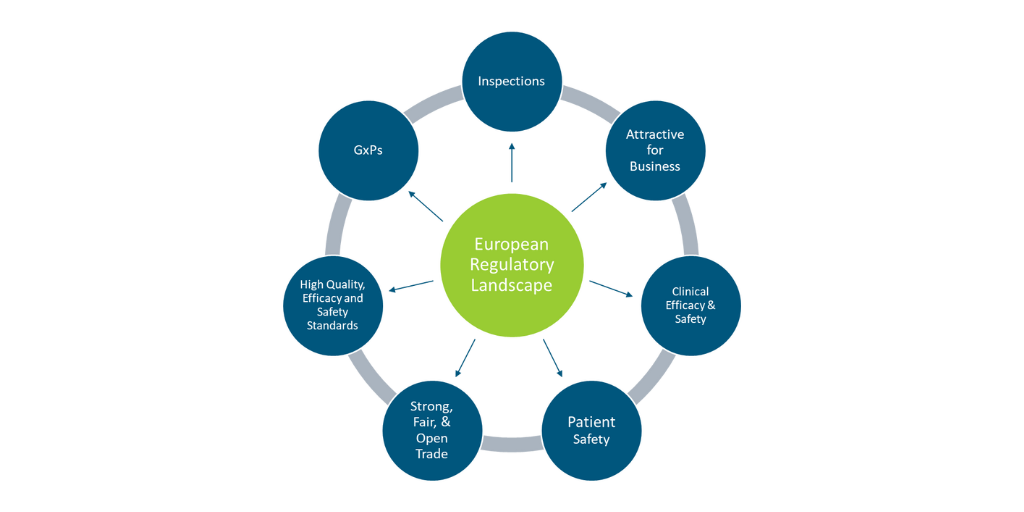
On 25 November 2020, the European Commission (EC) adopted the patient-centric New Pharmaceutical Strategy for Europe and published its roadmap for the revision of the general pharmaceutical legislation in 2021. The European Commission has begun initiating work in several areas, including the pediatric and orphan review.
In this article, you will get a comprehensive overview of the following:
Read the full article to learn more: https://www.regulatoryrapporteur.org/european-medicines-and-medical-devices-future-trends/222.article
2 min read
May 5, 2026 Veristat Events
May 29 - June 2, 2026 | Chicago, IL
Veristat is pleased to attend the ASCO...
1 min read
Apr 30, 2026 Veristat Events
May 12-13, 2026 – New Brunswick, NJ
Veristat is pleased to...